Patients Are the Why. Research Is the How.
FARE, the largest charitable supporter of food allergy research, is driving innovation toward a new, more promising day. A day when patients don’t have to endure an oral food challenge to get a reliable diagnosis. When families don’t need to pack epinephrine every time they leave the house. When kids can attend a birthday party or class trip without fear of an emergency room visit. Toward that day, FARE is advancing breakthroughs in food allergy prevention, diagnosis, treatment and care.
Elevating Care
Food allergy research and treatment are at a critical turning point. Emerging science and data point toward a new era in which new therapeutic options can help deliver the freedom and peace-of-mind that individuals with food allergy deserve. Through initiatives to develop innovative infrastructure for research, education and clinical care, FARE is committed to making effective, patient-centered approaches to prevention, diagnosis and treatment a reality.

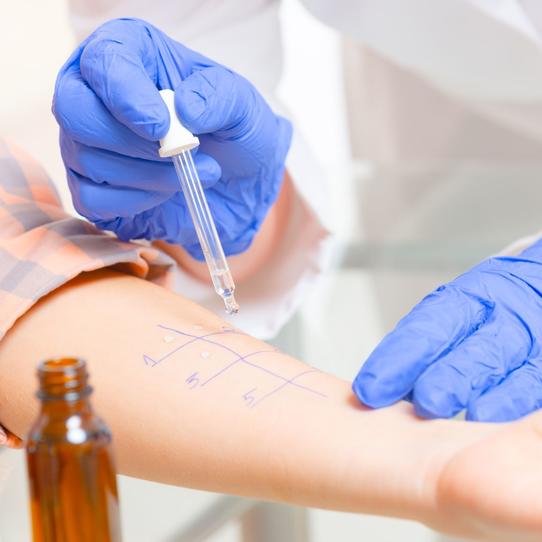
Pioneering Advances in Diagnosis
In an era of molecular diagnostics/medicine, food allergy diagnostics lag behind. FARE is currently working on driving research and innovation to bring new diagnostic methods for food allergy. Learn more here about methods of diagnosis currently in use, and why better tools are essential to improving the lives of individuals and families managing food allergy.
Early Introduction of Food Allergens
During the past decade, research findings have transformed the guidance that pediatricians and allergists and immunologists give to parents about when to introduce common food allergens to children at high risk for food allergies. The far-reaching results of this research show how parents, caregivers and health care providers can fight the food allergy epidemic.

Developing Effective Treatments
Researchers are investigating exciting new ways to stop food allergy reactions or limit reaction symptoms to someday create more effective, better-tolerated treatments that are accessible to all living with food allergies. We are on the cusp of revolutionary changes in the food allergy treatment landscape.

Support Food Allergy Research
Donate to Support FARE
Future In Sight with Peter Kolchinsky
In this monthly podcast, Peter engages with equally sharp and unfiltered geniuses about the potential of solving big picture problems in our lifetimes.
Your food allergy story powers research that changes lives.
Join the FARE Patient Registry® - making a difference is easy and confidential.
Join Patient RegistryRecent FARE-Funded Research

Information for Researchers
FARE is the largest private source of funding for food allergy research and has invested over $100 Million to date. See more about current initiatives and grants for the research community.


